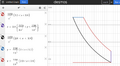
Diesel Cycle
Introduction
-
Diesel Cycle
-
Diesel Cycle
Ratio of specific heats (heat capacity ratio) is defined as
- Isentropic (adiabatic) expansion
- Isochoric cooling (Qout): Heat rejection. Power stroke ends, heat rejection starts.
- Isobaric compression: Exhaust
- Isobaric expansion: Intake
- Isentropic (adiabatic) compression
- Isobaric heating (Qin): Combustion of fuel (heat is added in a constant pressure;)
Engine displacement is the cylinder volume swept by all of the pistons of a piston engine, excluding the combustion chambers. A combustion chamber is part of an internal combustion engine in which the fuel/air mix is burned.
Only air is compressed, and then diesel fuel is injected directly into that hot, high-pressure air.
- Cylinder pressure: ~30–80 bar
- Injection pressure: ~1,000–2,500+ bar
Realistic Diesel Cycle
-
The size of the combustion chamber of MB W211.
Ratio of specific heats γ
Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \gamma = \frac{ \sum y_1 c_{p,i} }{ \sum y_1 ( c_{p,i} -R_u ) } }
| Condition | γ (approx.) |
|---|---|
| Stoichiometric | ~1.30–1.33 |
| Moderate lean | ~1.33–1.37 |
| Very lean (diesel) | ~1.37–1.40 |
Injection pressures
- Older systems: 200–500 bar
- Modern common-rail: 1,000–2,500 bar
- Latest systems: up to ~3,000 bar
Diesel-air Mixture
The heat capacity ratio (known as the adiabatic index) for a diesel-air mixture is typically around 1.4.
| 14.5:1 | Near-stoichiometric; good combustion efficiency but higher emissions. |
| 16:1 | Balanced performance; good power output and efficiency. |
| 18:1 | Lean burn; improved fuel economy but potential for higher NOx emissions. |
Diesel is a complicated compound, but it’s commonly approximated as a hydrocarbon like C12H23 (or C12H26). Air is about 21% O2 and 79% N2. Without nitrogen, the stoichiometric ratio is about
C12H23+17.75O2→12CO2+11.5H2O
and by including nitrogen, we get
C12H23+17.75( O2 + 3.76 N2 )→12CO2+11.5H2O + 66.74 N2.
The molar masses
- Fuel: 167 g/mol
- Air: 137.28 g/mol
And the total air needed is 17.72 x 137.28 = 2436 g, which gives air-to-fuel-ratio to
Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle AFR = \frac{2436}{167} = 14.6:1 }
MB data
Mercedes Benz W211 (2003)
- Engine displacement: 3222 cm3 = 0.003222 m3
- Bore x Stroke: 88.0 x 88.4 mm3
- Compression Ratio: 18.0
Bore x stroke gives V = 6xπ(8.8/2)2 x 8.84 cm3 = 3225.95cm3, which is rather close.
Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle p_1 V_1^\gamma = \text{constant}_1 }