Carnot Cycle
From wikiluntti
Introduction
- Isothermal expansion (No heat transfer / energy transfers): Heat is transferred from the hot reservoir to the gas.
- Isentropic (reversible adiabatic: Heat transfers / no energy transfer) expansion: without transfer of heat to or from a system, so that Q = 0, is called adiabatic, and such a system is said to be adiabatically isolated. Eg. the compression of a gas within a cylinder of an engine is assumed to be rapid that little of the system's energy is transferred out as heat to the surroundings.
- Isothermal compression
- Isentropic compression
Ideal Gas
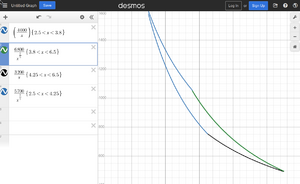
, or more generally polytropic process: , where is different processes depending on the value of the n:
- n = 0: isobaric
- n = ∞: isochoric
- n = 1: isothermal
- n = γ: isentropic (adiabatic)
Adiabatic index is for the air 7/5. For the ideal gas we have and .
- (n=1) Isothermal compression: T is constant, thus we have .
- (n=γ) Isentropic






