Diesel Cycle: Difference between revisions
| Line 73: | Line 73: | ||
Which are widely used in CFD and thermodynamics. | Which are widely used in CFD and thermodynamics. | ||
Compressibility factor Z (real gas correction). If you want a generalized ideal gas, you often introduce <math>p=Z\rho RT</math>. Where | |||
* Z=1 → ideal gas | * Z=1 → ideal gas | ||
* Z≠1 → real gas behavior | * Z≠1 → real gas behavior | ||
| Line 81: | Line 81: | ||
<math> | <math> | ||
Z=1+\frac{B(T)}V+\frac{C(T)}{V^2}+ \cdots | Z=1+\frac{B(T)}V+\frac{C(T)}{V^2}+ \cdots | ||
\approx 1+ \frac{B(T)}{RT}p | \approx 1+ \frac{B(T)}{RT}p. | ||
</math> | </math> | ||
Mixture-based formulation (more fundamental). Air is a mixture mainly of: | Mixture-based formulation (more fundamental). Air is a mixture mainly of: | ||
* N₂ (~78%) | * N₂ (~78%) | ||
Revision as of 20:59, 30 March 2026
Introduction
1
-
Diesel Cycle
-
Diesel Cycle
Ratio of specific heats (heat capacity ratio) is defined as
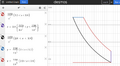
pV diagram
- Isentropic (adiabatic) expansion
- Isochoric cooling (Qout): Heat rejection. Power stroke ends, heat rejection starts.
- Isobaric compression: Exhaust
- Isobaric expansion: Intake
- Isentropic (adiabatic) compression
- Isobaric heating (Qin): Combustion of fuel (heat is added in a constant pressure;)
Engine displacement is the cylinder volume swept by all of the pistons of a piston engine, excluding the combustion chambers. A combustion chamber is part of an internal combustion engine in which the fuel/air mix is burned.
Only air is compressed, and then diesel fuel is injected directly into that hot, high-pressure air.
- Cylinder pressure: ~30–80 bar
- Injection pressure: ~1,000–2,500+ bar
Diesel Cycle and Ideal Gas
For a closed system, the total change in energy of a system is the sum of the work done and the heat added , and the reversible work done on a system by changing the volume is .
If the system is reversible and adiabatic (isentropic) , which gives
Furthermore, for any transformation of an ideal gas, it is always true that , giving
Real gas: Air
Basic ideal gas model (good first approximation). Air is usually approximated as an ideal gas with:
- Gas constant:
- R≈287 J/(kg\cdotpK)
- R≈287J/(kg\cdotpK)
- Equation of state:
- p=ρRT
This works well at:
- pressures near atmospheric
- temperatures roughly 200–500 K
“Generalized” ideal gas → temperature-dependent properties. To go beyond the simple model, you allow properties like heat capacity to vary with temperature:
Heat capacity. For air, a common approximation is a polynomial Typical coefficients (for dry air, ~200–1000 K range):
- a≈1005
- b≈0.1
More accurate forms come from NASA polynomials: Which are widely used in CFD and thermodynamics.
Compressibility factor Z (real gas correction). If you want a generalized ideal gas, you often introduce . Where
- Z=1 → ideal gas
- Z≠1 → real gas behavior
For air:
- At normal conditions: Z≈1
- At high pressure: use virial expansion
Mixture-based formulation (more fundamental). Air is a mixture mainly of:
- N₂ (~78%)
- O₂ (~21%)
- Ar (~1%)
Realistic Diesel Cycle
-
The size of the combustion chamber of MB W211.
Ratio of specific heats γ
| Condition | γ (approx.) |
|---|---|
| Stoichiometric | ~1.30–1.33 |
| Moderate lean | ~1.33–1.37 |
| Very lean (diesel) | ~1.37–1.40 |
Injection pressures
- Older systems: 200–500 bar
- Modern common-rail: 1,000–2,500 bar
- Latest systems: up to ~3,000 bar
- Air pressure in cylinder: ~30–80 bar, which is the pressure of the combustion chamber.
- Temperature: ~700–1000 K
Diesel-air Mixture
Only diesel is ejected into the cylinder. Clean air comes in during the intake stroke.
The heat capacity ratio (known as the adiabatic index) for a diesel-air mixture is typically around 1.4.
| 14.5:1 | Near-stoichiometric; good combustion efficiency but higher emissions. |
| 16:1 | Balanced performance; good power output and efficiency. |
| 18:1 | Lean burn; improved fuel economy but potential for higher NOx emissions. |
Diesel is a complicated compound, but it’s commonly approximated as a hydrocarbon like C12H23 (or C12H26). Air is about 21% O2 and 79% N2. Without nitrogen, the stoichiometric ratio is about
C12H23+17.75O2→12CO2+11.5H2O
and by including nitrogen, we get
C12H23+17.75( O2 + 3.76 N2 )→12CO2+11.5H2O + 66.74 N2.
The molar masses
- Fuel: 167 g/mol
- Air: 137.28 g/mol
And the total air needed is 17.72 x 137.28 = 2436 g, which gives air-to-fuel-ratio to
MB data
Motor: OM648.
Mercedes Benz W211 (2003)
- Engine displacement: 3222 cm3 = 0.003222 m3
- Bore x Stroke: 88.0 x 88.4 mm3
- Compression Ratio: 18.0
Bore x stroke gives V = 6xπ(8.8/2)2 x 8.84 cm3 = 3225.95cm3, which is rather close.